You need to agree to share your contact information to access this model
This repository is publicly accessible, but you have to accept the conditions to access its files and content.
You agree to not use the model to conduct experiments that cause harm to human subjects.
Log in or Sign Up to review the conditions and access this model content.

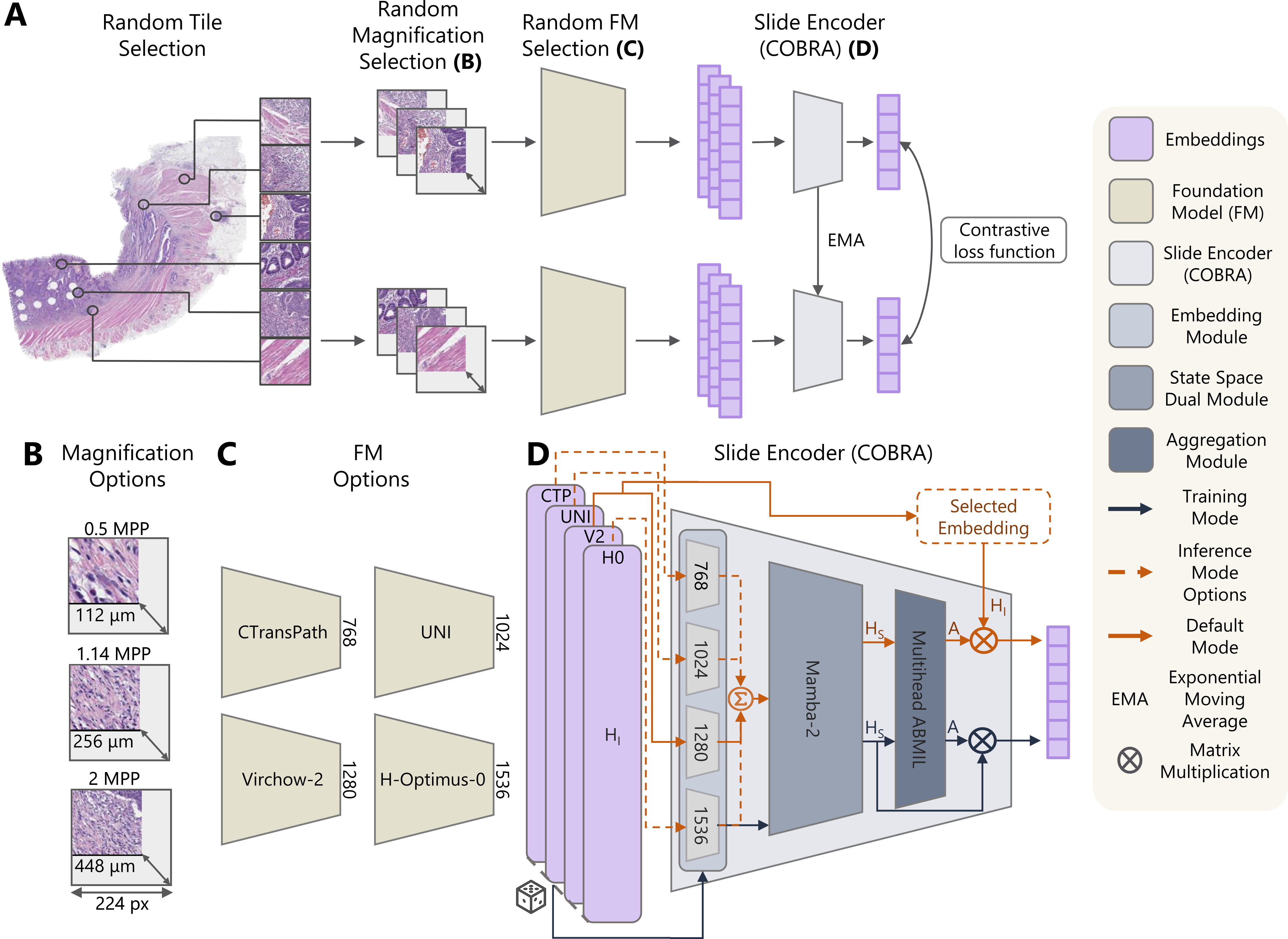
COntrastive Biomarker Representation Alignment (COBRA)
Abstract
Representation learning of pathology whole-slide images (WSIs) has primarily relied on weak supervision with Multiple Instance Learning (MIL). This approach leads to slide representations highly tailored to a specific clinical task. Self-supervised learning (SSL) has been successfully applied to train histopathology foundation models (FMs) for patch embedding generation. However, generating patient or slide level embeddings remains challenging. Existing approaches for slide representation learning extend the principles of SSL from patch level learning to entire slides by aligning different augmentations of the slide or by utilizing multimodal data. By integrating tile embeddings from multiple FMs, we propose a new single modality SSL method in feature space that generates useful slide representations. Our contrastive pretraining strategy, called COBRA, employs multiple FMs and an architecture based on Mamba-2. COBRA exceeds performance of state-of-the-art slide encoders on four different public Clinical Protemic Tumor Analysis Consortium (CPTAC) cohorts on average by at least +4.4% AUC, despite only being pretrained on 3048 WSIs from The Cancer Genome Atlas (TCGA). Additionally, COBRA is readily compatible at inference time with previously unseen feature extractors. Code available at https://github.com/KatherLab/COBRA.
News
- [Feb 27th 2025] Our paper has been accepted to CVPR 2025! 🎉
- [Feb 7th 2025]: COBRA II trained on all TCGA cohorts, is now live and ready to use!!
Installation
To install the necessary dependencies, run the following commands:
git clone https://github.com/KatherLab/COBRA.git && cd COBRA
pip install uv
uv venv --python=3.11
source .venv/bin/activate
uv pip install torch==2.4.1 setuptools packaging wheel numpy==2.0.0
uv sync --no-build-isolation
If there are any issues, consider also installing hatchling and editables:
uv pip install hatchling editables
And make sure python3.11-devel (or python3.11-dev) is installed. For Fedora or derivatives:
dnf install python3.11-devel
For Debian or derivatives:
apt install python3.11-dev
WSI Level Embeddings
To deploy the COBRA model, follow these steps:
Prepare your data: Extract tile embeddings with one or more patch encoders of your choice using STAMP.
- COBRA I:
- Supported tissue types: LUAD, LUSC, STAD, CRC, BRCA
- Supported patch encoders to generate weighting: CTransPath, UNI, Virchow2, H_optimus_0
- Supported patch encoders for patch feature aggregation: all existing patch encoders
- COBRA II:
- Supported tissue types: all cohorts included in TCGA
- Supported patch encoders to generate COBRAII weighting: CONCH, UNI, Virchow2, H_optimus_0
- Supported patch encoders for patch feature aggregation: all existing patch encoders
- COBRA I:
Request Access on Huggingface.
Extract COBRA Features:
The extraction scripts allow you to obtain slide‑ or patient‑level embeddings. In addition to standard command‑line arguments, you can now supply a YAML configuration file (using the--configflag) which overrides or specifies all extraction parameters (such as input directories, checkpoint paths, top‑k selection, etc.).Example configuration (extract_feats_config.yml):
extract_feats: download_model: false checkpoint_path: "/path/to/checkpoint.pth.tar" top_k: 100 output_dir: "/path/to/extracted/output" feat_dir: "/path/to/tile_embeddings" feat_dir_a: "/path/to/tile_embeddings_aux" # Optional, for aggregation features model_name: "COBRAII" patch_encoder: "Virchow2" patch_encoder_a: "Virchow2" h5_name: "cobra_feats.h5" microns: 224 slide_table: "/path/to/slide_table.csv" # Provide for patient-level extraction, omit for slide-levelUsage:
Slide-level extraction (without a slide table):
python -m cobra.inference.extract_feats_slide --feat_dir "/path/to/tile_embeddings" --output_dir "/path/to/slide_embeddings" --checkpoint_path "/path/to/checkpoint.pth.tar"Or by providing a configuration file:
python -m cobra.inference.extract_feats_slide --config /path/to/extract_feats_config.ymlPatient-level extraction (using a slide table):
python -m cobra.inference.extract_feats_patient --feat_dir "/path/to/tile_embeddings" --output_dir "/path/to/patient_embeddings" --slide_table "/path/to/slide_table.csv" --checkpoint_path "/path/to/checkpoint.pth.tar"Or with configuration:
python -m cobra.inference.extract_feats_patient --config /path/to/extract_feats_config.yml
Note: You have the option of providing different directories for weighting and aggregation steps. The script will load primary features from
--feat_dirand, if provided, additional features from--feat_dir_a. Features are matched by their coordinates before aggregation.
Using Extracted COBRA Features for Crossvalidation
After extracting the COBRA features (either at the slide or patient level), you can run crossvalidation to train and evaluate a downstream MLP classifier. The crossvalidation workflow is managed by two main scripts.
1. Training with Crossvalidation
The cobra/crossval/train.py script performs stratified K-fold crossvalidation and saves test predictions, AUROC scores, and the best model checkpoints per fold.
You need to supply a configuration file that specifies the following:
- CSV/Excel metadata file with patient IDs and target values.
- H5 file with extracted features.
- Output folder for saving checkpoints and results.
- Hyperparameters for training (learning rate, hidden dimension, batch size, number of folds, etc.).
Example configuration (crossval.yml):
train:
csv_path: "/path/to/metadata.csv"
target_column: "TARGET"
patient_id_column: "PATIENT_ID"
h5_path: "/path/to/extracted_features.h5"
output_folder: "/path/to/crossval/results"
hps:
lr: 0.0005
hidden_dim: 512
max_epochs: 64
patience: 16
batch_size: 32
num_workers: 8
n_folds: 5
dropout: 0.3
deploy:
csv_path: "/path/to/test_metadata.csv"
target_column: "TARGET"
patient_id_column: "PATIENT_ID"
h5_path: "/path/to/extracted_features.h5"
output_folder: "/path/to/deploy/results"
label_encoder_path: "/path/to/label_encoder.pkl"
hps:
hidden_dim: 512
n_folds: 5
Usage:
Train with:
python -m cobra.crossval.train -c /path/to/crossval.yml
During training, the script:
- Loads the CSV metadata and matches patient IDs with those in the H5 file.
- Encodes the target labels and saves the encoder.
- Splits data using Stratified K-Fold.
- Trains the MLP with PyTorch Lightning (using early stopping and checkpoint callbacks).
- Evaluates each fold and saves detailed results (including per-patient predictions and AUROC scores).
2. Deployment and Evaluation
The corresponding deployment script (cobra/crossval/deploy.py) lets you evaluate a trained model on unseen data. Its configuration file should include:
- CSV metadata file with test targets.
- H5 file with features used during inference.
- Path to the saved label encoder from training.
- Output folder for saving summaries and predictions.
Usage:
python -m cobra.crossval.deploy -c /path/to/crossval.yml
This script loads the best checkpoints from training, matches test patient IDs with the H5 file, computes evaluation metrics (e.g., AUROC), and saves both per-fold and aggregated results.
Note: Use your crossvalidation configuration file to supply all necessary file paths and hyperparameters.
Generating Heatmaps
The cobra/inference/heatmaps.py script generates visual heatmaps of WSIs by overlaying model attention maps.
How It Works
- Reads tile feature files (HDF5) and corresponding WSIs.
- Computes attention values from the COBRA model.
- Creates a composite image combining the slide thumbnail and a heatmap.
- Adds a 2 mm scale bar for reference.
- Saves the final composite as a PDF.
Example Configuration (heatmap_config.yml)
heatmap:
feat_dir: "/path/to/tile_embeddings" # Directory for tile feature files (HDF5)
wsi_dir: "/path/to/wsi_files" # Directory for whole slide images
checkpoint_path: "/path/to/checkpoint.pth.tar" # Model checkpoint path
microns: 112 # Microns per patch used for extraction
patch_size: 224 # Size of each patch in pixels
output_dir: "/path/to/heatmap/output" # Where to save generated heatmaps
stamp_version: 2 # Stamp version used during extraction
Usage
- With a configuration file:
python -m cobra.inference.heatmaps -c /path/to/heatmap_config.yml
- Or via command-line arguments:
python -m cobra.inference.heatmaps \
-f "/path/to/tile_embeddings" \
-s "/path/to/wsi_files" \
-w "/path/to/checkpoint.pth.tar" \
-r 112 \
-p 224 \
-o "/path/to/heatmap/output" \
-v 2
Pretraining via Self-Supervised Learning (SSL)
COBRA also supports pretraining using a self-supervised learning (SSL) framework adapted from MoCo-v3.
What It Does
- Pretrains a COBRA-based model using tile-level features.
- Uses a YAML configuration to specify model parameters, data paths, and training hyperparameters.
- Saves pretrained weights for later use in feature extraction and downstream tasks.
Example Configuration (cobraII.yml)
model:
nr_heads: 4
nr_mamba_layers: 1
dim: 768
input_dims:
- 512
- 1024
- 1280
- 1536
l_dim: 256
att_dim: 256
dropout: 0.2
d_state: 128
model_name: "cobraII"
ssl:
moco_m: 0.99
moco_t: 0.2
lr: 5e-4
warmup_epochs: 50
weight_decay: 0.1
epochs: 2000
workers: 56
batch_size: 1024
general:
nr_feats: 768
fms:
- "fm1"
- "fm2"
- "fm3"
- "fm4"
feat_base_paths:
- "/path/to/features_set1"
- "/path/to/features_set2"
paths:
out_dir: "/path/to/pretrain/output"
Usage
python -m cobra.ssl.pretrain -c /path/to/config.yml
This script sets up the SSL pretraining using your specified configuration and trains the model on tile features.
References
- CTransPath
Xiyue Wang, Sen Yang, Jun Zhang, Minghui Wang, Jing Zhang, Wei Yang, Junzhou Huang, and Xiao Han. Transformer-based unsupervised contrastive learning for histopathological image classification. Medical Image Anal- ysis, 2022
- UNI
Richard J Chen, Tong Ding, Ming Y Lu, Drew FK Williamson, Guillaume Jaume, Bowen Chen, Andrew Zhang, Daniel Shao, Andrew H Song, Muhammad Shaban, et al. Towards a general-purpose foundation model for com- putational pathology. Nature Medicine, 2024
- Virchow2
Eric Zimmermann, Eugene Vorontsov, Julian Viret, Adam Casson, Michal Zelechowski, George Shaikovski, Neil Tenenholtz, James Hall, David Klimstra, Razik Yousfi, Thomas Fuchs, Nicolo Fusi, Siqi Liu, and Kristen Sever- son. Virchow2: Scaling self-supervised mixed magnification models in pathology, 2024
- H-Optimus-0
Charlie Saillard, Rodolphe Jenatton, Felipe Llinares-López, Zelda Mariet, David Cahané, Eric Durand, and Jean-Philippe Vert. H-optimus-0, 2024
- CONCH
Lu, Ming Y. and Chen, Bowen and Zhang, Andrew and Williamson, Drew F. K. and Chen, Richard J. and Ding, Tong and Le, Long Phi and Chuang, Yung-Sung and Mahmood, Faisal. A visual-language foundation model for computational pathology. Nature Medicine, 2024
- STAMP
Omar S. M. El Nahhas, Marko van Treeck, Georg Wölflein, Michaela Unger, Marta Ligero, Tim Lenz, Sophia J. Wagner, Katherine J. Hewitt, Firas Khader, Sebastian Foersch, Daniel Truhn, and Jakob Nikolas Kather. From whole-slide im- age to biomarker prediction: end-to-end weakly supervised deep learning in computational pathology. Nature Protocols, 2024
- MoCo-v3
Xinlei Chen*, Saining Xie*, and Kaiming He. An empirical study of training self-supervised vision transformers. arXiv preprint arXiv:2104.02057, 2021
Citation
If you find our work useful in your research or if you use parts of our code please consider citing our paper:
@inproceedings{cobra,
title = {Unsupervised Foundation Model-Agnostic Slide-Level Representation Learning},
author = {Tim Lenz* and Peter Neidlinger* and Marta Ligero and Georg Wölflein and Marko van Treeck and Jakob Nikolas Kather},
booktitle = {Proceedings of the IEEE/CVF Conference on Computer Vision and Pattern Recognition (CVPR)},
year = {2025}
}


